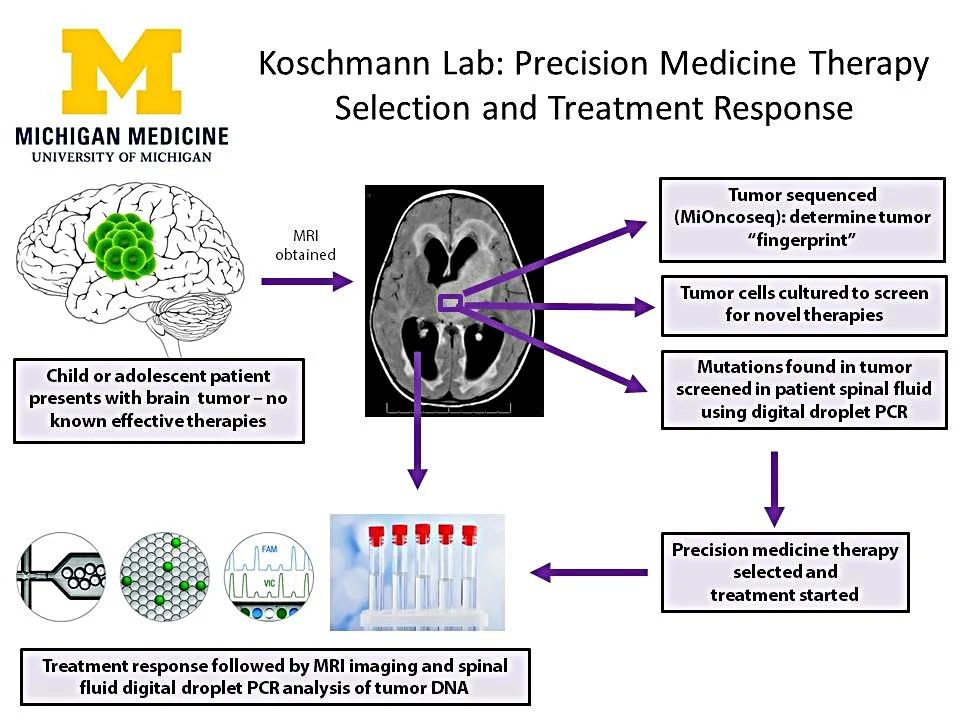
The Koschmann lab is applying a novel precision medicine approach accounting for both unique tumor biology and optimization of blood brain barrier penetration. We are performing correlate biology work on ongoing precision medicine trials for children and young adults with brain tumors at the University of Michigan, including correlation of spinal fluid analysis with surveillance MRIs.
Serial H3K27M cell-free tumor DNA (cf-tDNA) tracking predicts ONC201 treatment response and progression in diffuse midline glioma
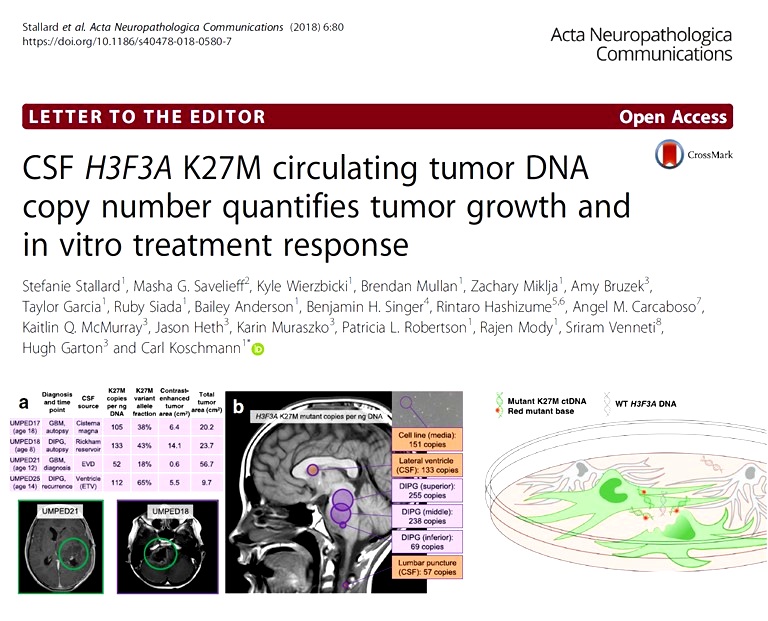
We reported (Cantor et al Neuro Oncology. 2022,) the first demonstrated feasibility for serial CSF collection in a prospective high-grade glioma clinical trial. Nonrecurrent patients (those enrolled after initial radiation) with a decrease in CSF H3.3K27M variant allele fraction (VAF) displayed a doubling of time to progression. “Spikes” in cf-tDNA VAF (increase of at least 25%) preceded progression in many cases. In individual cases, patterns of change in VAF over time demonstrate additional clinical utility in terms of differentiating pseudo-progression and bevacizumab (“pseudo-response”) effect. These findings represent a new direction for understanding the feasibility and utility of H3K27M cf-tDNA as a clinically relevant prognostic marker.